bioPROTAC and transcription factors
Intrinsically-disordered proteins are a hard paradigm for the typically-designed types of protein degrader medicines. Intrinsically-

PROTAC vs. bioPROTAC
Targeted Protein degradation began by accident – with tragedy of medicines in pregnancy. The resulting birth defects caused by Thalidomide informed researchers that a transcription factor (SALL4) could be brought in proximity to protein recycling machinery (CRBN) and lead to elimination of the transcription factor. The role of Thalidomide as a Molecular Glues inspired other such glues, but also artificial ones – with PROTACs (PROteolysis TArgeting Chimeras) as a prototype.
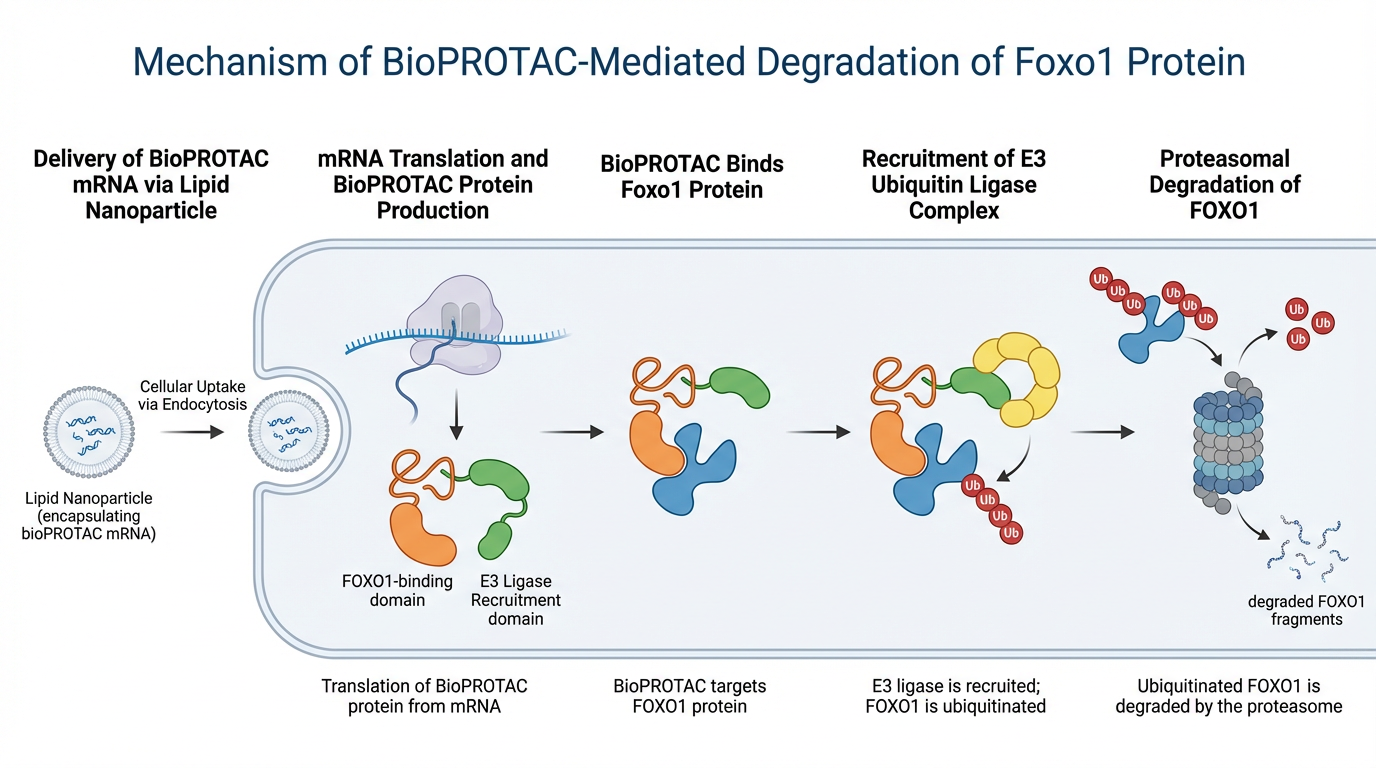
However, a PROTAC relies on a small chemical “hook” to bring the protein intended for degradation. For proteins that do not reach a steady-state form (shape), but instead move freely, a small chemical hook with tight binding can be elusive. Nature, however, offers a solution to binding proteins that are intrinsically-disorder – namely, other proteins such as antibodies and nanobodies.
This is where bioPROTACs provide a key advantage: instead of relying on small-molecule binding, they use engineered protein binders to achieve high specificity and strong interaction even with flexible, disordered targets. As a result, bioPROTACs can access a much broader range of disease-driving proteins—particularly those considered “undruggable”—making targeted protein degradation both more precise and more versatile.