ADC Antibody Drug Conjugation
Antibody–Drug Conjugates (ADCs) are an advanced class of cancer therapeutics that combine the precision of targeted therapy with the potency of chemotherapy. They are designed to selectively deliver powerful anti-cancer drugs directly to tumor cells while limiting damage to healthy tissue, making them a key innovation in modern oncology.
Antibody Drug Conjugation
Deliver poison selectively inside cancer cells
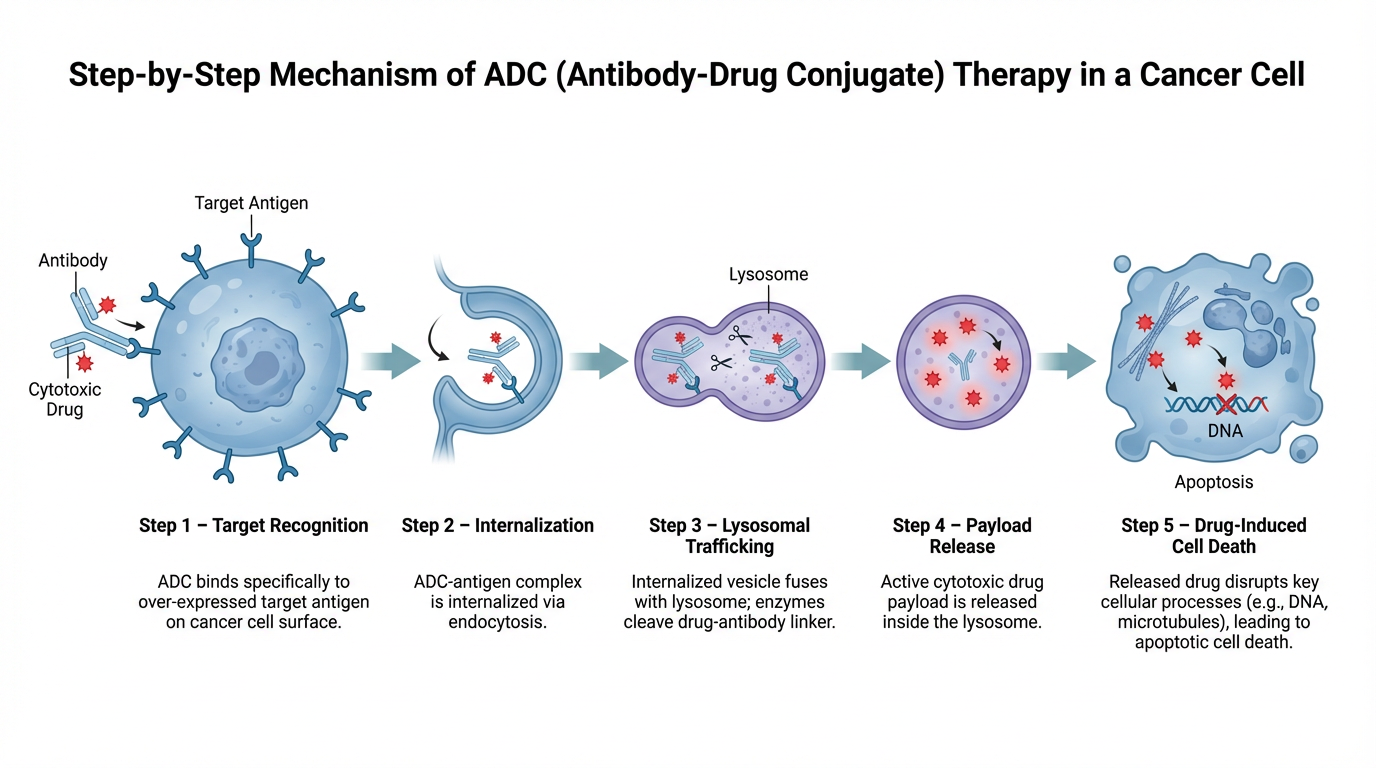
An ADC consists of three main components: a monoclonal antibody, a cytotoxic payload, and a linker that connects them. The antibody is engineered to recognize a specific antigen that is highly expressed on cancer cells. This targeting mechanism allows the ADC to bind selectively to tumor cells, acting like a guided delivery system. Once bound, the ADC is internalized into the cancer cell through receptor-mediated endocytosis. Inside the cell, the linker is broken down, releasing the cytotoxic drug. These payloads are extremely potent and work by disrupting critical cellular functions such as DNA replication or microtubule formation, ultimately causing cell death.
A major advantage of ADCs is their ability to use drugs that would be too toxic if given systemically. Traditional chemotherapy affects both healthy and cancerous cells, leading to significant side effects. ADCs, by contrast, concentrate their effects within tumor cells, improving the balance between efficacy and safety. Each component of an ADC must be carefully optimized. The target antigen should be abundant on cancer cells but limited on normal tissues to reduce off-target effects. The antibody must bind strongly and specifically to this antigen, while the linker must remain stable in the bloodstream but release the drug efficiently once inside the cell.
The payload is typically a highly potent small molecule, often a microtubule inhibitor or DNA-damaging agent. Because only a few drug molecules can be attached to each antibody, these payloads must be effective at very low concentrations. ADCs have already shown success in treating several cancers, including breast cancer and lymphomas, and many more are in development. Their modular design allows researchers to adapt them for different cancer types by swapping out the antibody, linker, or payload.
However, challenges remain. These include potential toxicity if the drug is released too early, and resistance mechanisms where cancer cells reduce antigen expression or alter drug processing. Manufacturing is also complex, requiring precise control to ensure consistent quality and performance. Ongoing research is focused on improving ADC design through more stable linkers, novel payloads, and better targeting strategies. Some next-generation ADCs are also being developed to stimulate the immune system, opening the door for combination therapies with immunotherapy.
Overall, ADCs represent a powerful shift toward more precise and effective cancer treatment. By combining targeted delivery with high potency, they offer a promising path forward in the fight against cancer.